In a comprehensive study published in the journal Aging Cell, a team of researchers has uncovered a critical molecular mechanism that governs the structural integrity of the skin and the vitality of its regenerative stem cell populations. The research highlights the pivotal role of a dermal protein known as fibulin-5 (FBLN5) in maintaining the "fast-cycling" stem cells responsible for the rapid turnover and repair of the epidermis. This discovery offers new insights into why skin thins and loses its regenerative capacity as humans age, potentially opening doors for novel therapeutic interventions in dermatology and geriatric medicine.
The Cellular Architecture of the Epidermis
The human skin is a complex, multi-layered organ that serves as the body’s primary barrier against environmental pathogens, UV radiation, and physical trauma. To maintain this barrier, the outermost layer, the epidermis, must constantly renew itself. This renewal process is driven by two distinct populations of basal stem cells: slow-cycling cells and fast-cycling cells.
Slow-cycling stem cells are characterized by the expression of the protein DLX1. These cells act as a long-term reservoir, dividing infrequently to preserve the stem cell pool. In contrast, fast-cycling stem cells, identified by the expression of SLC1A3, are the "workhorses" of skin regeneration. They divide rapidly to provide a steady supply of new keratinocytes to replace those lost at the surface.
As biological aging progresses, the balance between these two populations shifts. Specifically, the population of fast-cycling SLC1A3-expressing cells begins to dwindle. This decline is a hallmark of skin aging, leading to a diminished ability to repair wounds and a progressive thinning of the epidermal layer. Until recently, the exact environmental cues within the skin that trigger this decline remained poorly understood.
The Role of the Extracellular Matrix and Fibulin Proteins
The behavior of skin stem cells is not determined solely by internal genetic programming; it is heavily influenced by the "niche" or environment in which they reside. This environment is largely defined by the extracellular matrix (ECM), a complex network of proteins and carbohydrates that provides structural support and biochemical signaling to surrounding cells.
The ECM is well-known to undergo significant degradation during aging. Key components like collagen and elastin break down, and the signaling receptors known as integrins—which allow cells to "sense" their environment—decline in number and efficiency. Recent research has pointed toward the fibulin family of proteins as essential regulators of this niche. Fibulin-7, for instance, was previously identified as a crucial factor for the proliferation of fast-cycling cells.
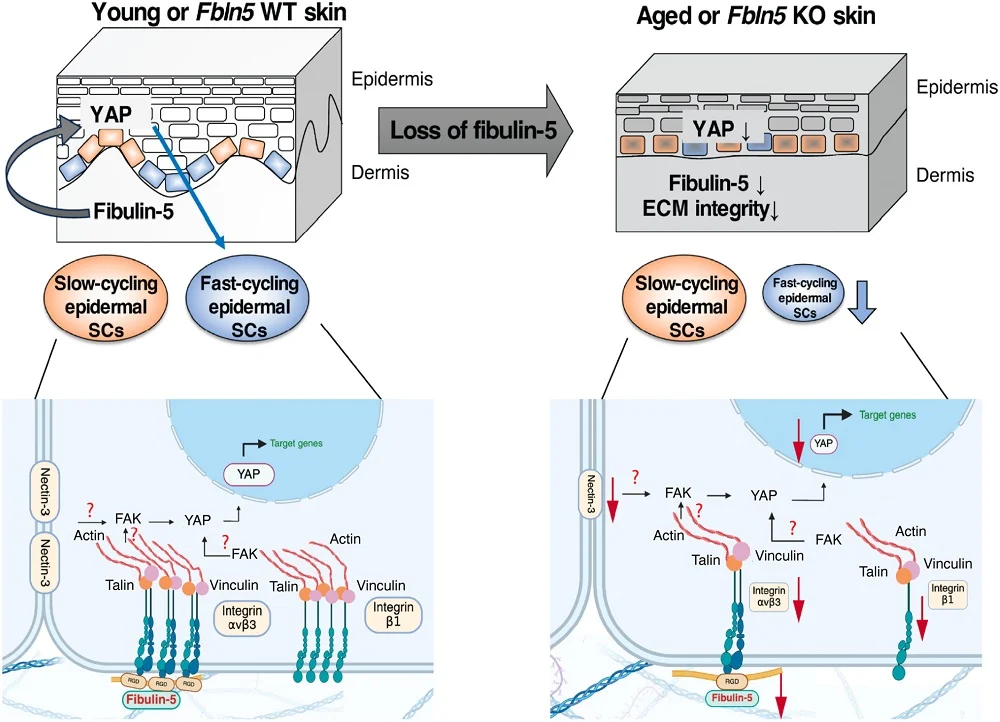
The current study focuses on fibulin-5, a related protein known for its role in elastic fiber assembly. Fibulin-5 is found in the dermis and has been documented to interact directly with integrins. Because fibulin-5 levels are known to decrease with age and UV exposure, researchers hypothesized that its loss might be a primary driver behind the disappearance of fast-cycling epidermal stem cells.
Experimental Findings: The Fibulin-5 Knockout Model
To test this hypothesis, the research team utilized a mouse model in which the gene for fibulin-5 was deactivated (knocked out). The physical results were striking and immediate. Even at a young age, the fibulin-5 deficient mice exhibited phenotypic traits usually reserved for much older wild-type mice. Their skin was noticeably loose and fragile, their black coats turned a duller brown, their hair became sparse, and they maintained a lower overall body weight.
By the time these modified mice reached 12 months of age—middle age for a mouse—the thinning of their skin was severe compared to their wild-type counterparts. Microscopic analysis of the skin architecture revealed a significant reduction in "scale" areas. In mouse biology, the tail skin is often used as a model where "scale" and "interscale" regions house fast-cycling and slow-cycling cells, respectively. In the knockout mice, the scale areas (the homes of the fast-cycling cells) were drastically diminished, mirroring the cellular landscape of natural senescence.
Genomic Alterations and Inflammaging
Beyond physical appearance, the researchers conducted a deep dive into the genetic profile of the skin in fibulin-5 deficient mice. They discovered a widespread downregulation of genes essential for healthy skin maintenance. These included genes involved in DNA replication, cellular adhesion (how cells stick to one another and the ECM), and the formation of the extracellular matrix itself.
Conversely, the study found an upregulation of inflammatory cytokines and specific stress-signaling pathways. This shift represents a molecular state often referred to as "inflammaging"—a chronic, low-grade inflammation that accelerates tissue breakdown. The genomic profile of the young knockout mice was remarkably similar to that of naturally aged, 24-month-old wild-type mice, suggesting that the absence of fibulin-5 effectively fast-forwards the skin’s biological clock.
The researchers also noted a breakdown in the integrity of the dermal-epidermal junction (DEJ), the critical interface where the two main layers of skin meet. In both naturally aged mice and fibulin-5 knockout mice, there was a significant decrease in collagen XVII and specific integrins (α6 and β4). However, the knockout mice showed a unique decline in integrin β3, highlighting that fibulin-5 provides a specific, necessary signal that other ECM components cannot replicate.
The YAP Signaling Pathway: A Mechanistic Link
The study’s most significant mechanical revelation involves the YAP (Yes-associated protein) signaling pathway. YAP is a powerful regulator of cell proliferation and organ size; it acts as a mechanosensor, telling cells to divide when they are in a healthy, supportive environment.
The researchers found that YAP levels were significantly lower in the skin of fibulin-5 deficient mice, naturally aged mice, and even in skin cell samples derived from elderly human patients. To confirm the link between YAP and the fast-cycling state, the team treated healthy wild-type mice with verteporfin, a drug that inhibits YAP. The result was a sharp decline in SLC1A3-expressing fast-cycling cells.
Crucially, when the researchers took human skin cells and directly exposed them to purified fibulin-5 protein, they observed a resurgence in the expression of SLC1A3. This suggests that fibulin-5 acts as a positive regulator of the fast-cycling state by maintaining YAP signaling. In essence, fibulin-5 tells the stem cells that the environment is stable enough to support rapid division.
Chronology of Scientific Context
The identification of fibulin-5 as a key player in skin aging is the latest step in a decades-long effort to map the "aging niche."
- 2005: Research established that fibulin-5 levels drop significantly in human skin due to both chronological aging and UV damage.
- 2010: Studies identified that integrin β1 and other stem cell markers decline in aging human skin.
- 2016: Scientists successfully mapped the lineage of DLX1 (slow-cycling) and SLC1A3 (fast-cycling) cells in the epidermis.
- 2022: Research into fibulin-7 showed its role in stem cell heterogeneity.
- Present: This new study in Aging Cell connects fibulin-5 to the YAP pathway and the specific loss of fast-cycling populations across species.
Implications for Future Medicine and Cosmetics
While the researchers have not yet performed "rescue" experiments—such as injecting fibulin-5 into living aged mice or using gene therapy to overexpress YAP—the implications are profound. If the loss of fast-cycling stem cells is a primary cause of skin thinning and poor wound healing, then restoring the fibulin-5/YAP axis could be a viable strategy for skin rejuvenation.
In the clinical setting, this could lead to the development of topical treatments or injectable scaffolds designed to mimic the effects of fibulin-5. Such treatments would be particularly beneficial for elderly patients suffering from chronic non-healing wounds or skin tears, which are common complications of aged, thin skin.
Furthermore, the study provides a concrete molecular target for the cosmetic industry. While many "anti-aging" products claim to boost collagen, targeting the specific proteins that maintain the stem cell niche—like fibulin-5—may offer a more fundamental way to preserve youthful skin function rather than just appearance.
Conclusion and Scientific Outlook
The study titled "Fibulin-5 maintains the fast-cycling epidermal stem cell population via YAP signaling" provides a robust framework for understanding the interplay between the extracellular environment and stem cell behavior. By demonstrating that the loss of a single dermal protein can trigger a cascade of aging markers—from thinned skin to shifted cell populations—the researchers have highlighted the fragility of the skin’s regenerative system.
The findings underscore that aging is not merely the accumulation of cellular "wear and tear," but a regulated process influenced by the loss of specific signaling molecules. Future research will likely focus on whether these pathways can be safely reactivated in humans and whether similar "fibulin-dependent" niches exist in other organs, such as the lungs or large blood vessels, where fibulin-5 is also known to play a structural role. For now, fibulin-5 stands as a promising candidate in the ongoing quest to understand and mitigate the biological effects of time on the human body.