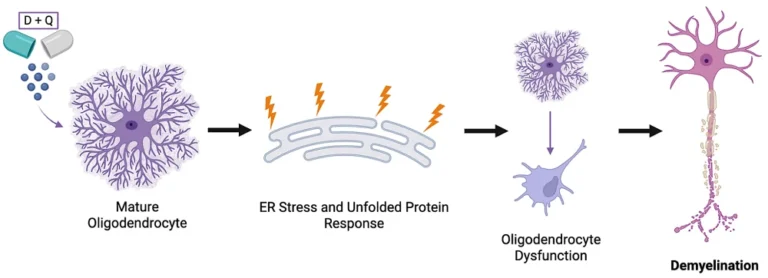
The pursuit of pharmaceutical interventions to arrest or reverse the biological aging process has encountered a significant complication following the publication of a new study in the Proceedings of the National Academy of Sciences (PNAS). Researchers have identified that the most prominent senolytic therapy—a combination of the leukemia drug dasatinib and the plant-derived flavonoid quercetin (D+Q)—induces cellular stress and demyelination in specific regions of the brain. These findings suggest that while senolytics remain a primary hope for extending human healthspan, their systemic application may carry neurodegenerative risks that mimic the pathological hallmarks of multiple sclerosis (MS).
For over a decade, the "geroscience hypothesis" has posited that by targeting fundamental aging processes, such as cellular senescence, scientists can delay the onset of multiple chronic diseases simultaneously. Senescent cells, often referred to as "zombie cells," are those that have ceased dividing due to damage or stress but refuse to die. Instead, they linger in tissues, secreting a pro-inflammatory cocktail known as the senescence-associated secretory phenotype (SASP). The D+Q combination became the "gold standard" for clearing these cells after early studies demonstrated its ability to improve physical function and lifespan in aged mice. However, the new data from the University of Connecticut School of Medicine and other contributing institutions suggests that the central nervous system may be uniquely vulnerable to the off-target effects of these compounds.
The Biological Role of Senescence in Brain Health
To understand the gravity of the study’s findings, it is necessary to examine the role of neural progenitor cells (NPCs) and oligodendrocytes in the brain. In a healthy brain, NPCs serve as a reservoir of stem cells capable of differentiating into various neural cell types, including oligodendrocytes. These oligodendrocytes are responsible for producing myelin, a fatty substance that wraps around axons to provide electrical insulation. This insulation is critical for the rapid transmission of nerve impulses and the metabolic support of neurons.
In neurodegenerative conditions such as multiple sclerosis, the immune system mistakenly attacks these myelin sheaths, leading to impaired cognitive and motor function. Previous research has established that in patients with progressive MS, a significant portion of NPCs become senescent. These senescent NPCs fail to mature into functional oligodendrocytes, thereby preventing the brain from repairing myelin damage. The logic behind the new study was to determine if clearing these senescent cells with D+Q could "unblock" the brain’s natural repair mechanisms.
Chronology of the Study and Experimental Design
The research team, led by Dr. Stephen J. Crocker and colleagues, utilized aged mouse models to simulate the environment of an aging human brain. The experimental protocol involved the oral administration of dasatinib and quercetin three times per week on alternating weeks—a dosage schedule previously shown in other studies to extend the median lifespan of mice and improve cardiovascular health.
One month after the initiation of the treatment, the researchers focused their analysis on the rostral corpus callosum (CC). The corpus callosum is the largest white matter structure in the brain, consisting of millions of axonal fibers that facilitate communication between the left and right cerebral hemispheres. Because of its high density of myelinated axons, the CC is a critical area for assessing the integrity of the brain’s white matter.
The researchers employed transmission electron microscopy (TEM) to achieve high-resolution visualization of the myelin structures. This allowed for the precise measurement of the "g-ratio," a standard metric used to determine the thickness of the myelin sheath relative to the diameter of the axon.
Unexpected Results: Demyelination and Morphological Retraction
The initial hypothesis suggested that D+Q would improve the brain environment by removing inflammatory senescent cells. Instead, the TEM analysis revealed a statistically significant reduction in myelination levels in the treated mice compared to the control group. Surprisingly, this effect was not limited to aged animals; follow-up experiments on young mice yielded similar results, indicating that the damage was not a result of pre-existing age-related vulnerability but was a direct consequence of the drug intervention.
Further investigation into the cellular mechanics revealed that D+Q was not killing the oligodendrocytes—the very cells it was meant to protect or support. Instead, the drugs appeared to induce a state of "functional paralysis." Within only 20 minutes of exposure to the treatment, the oligodendrocytes began to exhibit morphological changes. They retracted their complex branching outgrowths and reduced their deposition of myelin onto neighboring axons. This rapid response suggested a direct biochemical interference rather than a slow, cumulative toxic effect.
Identifying the Mechanism: Endoplasmic Reticulum Stress
To determine why the oligodendrocytes were failing, the researchers conducted a transcriptomic analysis to observe changes in gene expression. The data pointed toward a significant increase in endoplasmic reticulum (ER) stress. The ER is the cellular organelle responsible for the folding and assembly of proteins. When the ER is under stress, it triggers the Unfolded Protein Response (UPR). If the stress is not resolved, the cell’s ability to manufacture and export essential materials—such as the proteins required for myelin—is severely compromised.
In the case of the D+Q treated cells, the ER stress effectively silenced the machinery that controls myelin deposition. While the oligodendrocytes maintained their cellular identity and did not undergo apoptosis (programmed cell death), they were no longer capable of organizing myelin correctly. This left the axons exposed and vulnerable, creating a physiological state remarkably similar to the early stages of demyelinating diseases.
Supporting Data and Comparative Analysis
The study’s findings align with a growing body of evidence suggesting that senolytics may have tissue-specific consequences. While dasatinib is an effective tyrosine kinase inhibitor used in cancer therapy, its lack of specificity means it can interfere with signaling pathways essential for the maintenance of healthy glial cells. Quercetin, though a natural antioxidant, can also influence a wide array of metabolic pathways that, in the context of the blood-brain barrier, may lead to unintended outcomes.
Data from the study highlighted that the reduction in myelin was "modest but significant." In a clinical context, even a modest reduction in myelin thickness in the corpus callosum can lead to measurable declines in processing speed, executive function, and motor coordination. The fact that these changes were observable across different age groups suggests that the risk profile of D+Q may be broader than previously assumed by the longevity community.

Reactions and Implications for the Scientific Community
The publication of these results has prompted a call for a more nuanced approach to anti-aging medicine. While clinical trials for D+Q continue for conditions such as idiopathic pulmonary fibrosis and chronic kidney disease, neuroscientists are urging caution regarding the drug’s impact on the central nervous system.
"These findings serve as a vital reality check for the field of senolytics," noted an independent researcher in regenerative medicine. "It underscores the reality that ‘senescent’ is not always a synonym for ‘disposable.’ In some contexts, the pathways we are inhibiting to kill senescent cells are the same pathways healthy cells need to function."
For researchers specializing in multiple sclerosis, the study provides a paradoxical silver lining. Because the D+Q treatment induces MS-like damage without killing the cells, it provides a new, controllable model for studying oligodendrocyte dysfunction. By understanding how to reverse the ER stress caused by D+Q, scientists may discover new ways to stimulate remyelination in MS patients.
Broader Impact and the Future of Longevity Research
The implications of this study extend beyond the laboratory and into the burgeoning "biohacking" and self-experimentation communities. Because dasatinib and quercetin are relatively accessible—dasatinib through prescription and quercetin as an over-the-counter supplement—some individuals have begun self-administering the "D+Q protocol" in hopes of achieving life extension. The PNAS study provides a stark warning that such interventions could potentially cause silent, neurological damage.
The future of the senolytic field likely lies in the development of "second-generation" treatments. These would include:
- Targeted Delivery Systems: Using nanoparticles or specialized ligands to ensure senolytic drugs only enter specific tissues or target only specific cell types, sparing healthy oligodendrocytes.
- Senomorphics: Instead of killing cells, these compounds would suppress the harmful SASP secretions without interfering with the cell’s structural functions.
- Tissue-Specific Screening: Ensuring that new longevity drugs are tested not just for systemic toxicity, but for their specific impact on the blood-brain barrier and white matter integrity.
In conclusion, while the D+Q combination remains a landmark discovery in the fight against aging, the revelation of its impact on the brain’s wiring emphasizes the complexity of the human organism. The journey toward a longer life must be balanced with the preservation of cognitive integrity, ensuring that the "added years" are not marred by the very neurodegenerative conditions that senolytics were designed to prevent. The scientific community must now pivot toward refining these tools, moving from the broad-spectrum "sledgehammer" approach of early senolytics to the precision "scalpel" required for safe human application.


